This article is the first in a two-part series on ongoing challenges using immunotherapy in special populations. The second part of this series focuses on elderly patients.
By Valérie Gounant, MD, and Elisabeth Quoix, MD, PhD
Posted: December 11, 2019
Since the beginning of the 21st century, there have been two major innovations in the treatment of advanced NSCLC: the use of targeted therapies and the use of immune checkpoint inhibitors (ICIs). These modalities have revolutionized outcomes for patients with metastatic disease. However, the U.S. Food and Drug Administration (FDA) approvals for atezolizumab, nivolumab, and pembrolizumab were based on the results of phase III clinical trials, which restricted enrollment to patients with a performance status (PS) of 0 or 1, leaving the thoracic oncology community wondering about optimal treatment for patients with poor PS.
The prevalence of poor PS (2-4) patients at time of diagnosis is as high as 34%.1 For patients with metastatic NSCLC and PS 3-4, there is no recommendation for chemotherapy, and best supportive care is the usual standard in the absence of a molecular target. Most patients with PS 3-4 die within 2 to 4 months of diagnosis.
However, trials dedicated to patients with advanced NSCLC who harbor oncogenic drivers, including activating mutations, have been performed in populations with such poor general condition,2,3 and safety and efficacy were consistent with results observed in patients with good PS (so-called Lazarus syndrome), leading to general acceptance of these agents independent of PS. These trials have profoundly changed clinical practice; now, if oncogene-addicted tumors are detected, these patients are treated with oncogene-specific tyrosine kinase inhibitors, regardless of PS.
Current Data
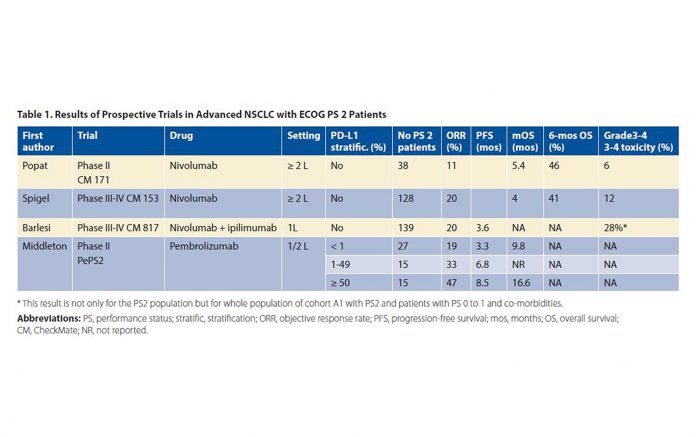
We have little data about safety and efficacy in poor PS patients. Currently, only four prospective trials including PS 2 patients have been published; three were in abstract form,4,5,6 and only one7 was an actual journal article. These included two phase II trials (CheckMate 171 with nivolumab4 and PePS2 with pembrolizumab5) and two phase III/IV trials (CheckMate 153 with nivolumab7 and CheckMate 817 with nivolumab and ipilimumab6). These trials did not select their populations based on biomarkers. The results of prospective trials in advanced NSCLC in PS 2 patients are summarized in Table 1 on page 3 (after Passaro8). These trials, apart from PePS2, also included elderly patients and /or PS 0-1 patients with comorbidities, making it difficult to draw specific conclusions for PS 2 patients, which already constitute a very heterogeneous population. Heightening heterogeneity by the inclusion of elderly patients and PS 0-1 patients with comorbidities introduces even more complexity and ambiguity in interpreting the results.
The incidence of grade 3 to 4 treatmentrelated adverse events (TRAE; primary endpoint) was 6% for the PS 2 population and 12% for the overall population (including PS 0-1 patients and patients aged 70 years and older) in CheckMate 171; comparable figures in CheckMate 153 were 9% for PS 2 and 6% for the overall population. In CheckMate 817, combination immunotherapy was more toxic, but the safety profi le was similar between cohort A with PS 0-1 patients (35% grade 3-4 TRAE) and cohort A1 with PS 2 patients (28% grade 3-4 TRAE) (139/198 patients) and PS 0-1 patients with comorbidities.
Overall survival (OS) was worse in PS 2 patients compared to the entire population in CheckMate 171 (5.4 vs. 9.9 months) and in CheckMate 153 (4 vs. 9.1 months). In CheckMate 817, in the PS 2 population, progression-free survival (PFS) was 3.6 months, with response rate (RR) of 20%, with median duration of response of 14.2 months. As expected, PFS was longer in those patients whose tumors had a high PD-L1 expression and/or high tumor mutation burden (TMB). In PePS2, RR was 19% in patients with PD-L1 expression of less than 1%, 33% in patients with PD-L1 expression of 1% to 49%, and 47% in patients with PD-L1 expression of 50% or higher. Median PFS and OS in those with PD-L1 expression of 50% or higher were 8.5 and 16.6 months, respectively. Thus, in selected populations, immunotherapy may be quite useful in PS 2 patients. This observation must be confirmed in dedicated studies confined to PS 2 patients, either pretreated or not, with or without the addition of chemotherapy. It is worth noting that, although all of the crucial phase III studies proving the benefits of ICIs were limited to PS 0-1 patients, FDA authorization for the use of ICIs do not restrict the use of these agents s to PS 0-1 patients.
Although there are no dedicated trials for PS 3 patients, we previously observed Lazarus-type responses to anti‒PD-1 ICIs in two patients with NSCLC in very poor condition but with very high PD-L1 expression.9 Such patients improved from PS 3 or higher before the initiation of immunotherapy, to PS 0, after only 1 month of ICIs; now after more than 24 months of follow-up, major tumor shrinkage continues. Recently, others have similarly suggested that pembrolizumab can be considered in critically ill patients with NSCLC and PD-L1 expression of 50% or more.10
Challenges and Potential Next Steps
The main challenge is to select poor PS populations who are more likely to derive a benefit from immunotherapy. In particular, it is necessary to define predictive biomarkers in this population: these include tumor biomarkers (e.g., molecular profile, PD-L1, and TMB) and patient biomarkers (e.g., inflammatory and nutritional markers). Several trials dedicated to PS 2 are currently recruiting. However, only two of these trials select the population based on PD-L1 status: these include the trial of the Swiss Group for Clinical Cancer Research (NCT03620669) and the SAVIMMUNE trial (NCT04108026) orchestrated by the French Cooperative Thoracic Intergroup (IFCT).
The role of chemotherapy and immunotherapy in combination must also be defined in the poor-PS population. A phase I trial evaluating the feasibility of weekly low-dose carboplatin and paclitaxel with pembrolizumab for patients with advanced NSCLC and PS 2-3 is ongoing.11 ✦
About the Authors: Dr. Gounant is with the Thoracic Oncology Department, Hôpital Bichat, APHP, France. Prof. Quoix is with the Department of Pneumology, University hospital of Strasbourg, France.
References:
1. Lilenbaum RC, Cashy J, Hensing TA, Young S, Cella D. Prevalence of poor performance status in lung cancer patients: implications for research. J Thorac Oncol 2008;3(2):125-9.
2. Inoue A, Kobayashi K, Usui K, Maemondo M, Okinaga S, Mikami I, et al. First-line gefitinib for patients with advanced non-small-cell lung cancer harboring epidermal growth factor receptor mutations without indication for chemotherapy. J Clin Oncol 2009;27(9):1394-400.
3. Iwama E, Goto Y, Murakami H, Harada T, Tsumura S, Sakashita H, et al. Alectinib for Patients with ALK Rearrangement-Positive Non- Small Cell Lung Cancer and a Poor Performance Status (Lung Oncology Group in Kyushu 1401). J Thorac Oncol 2017;12(7):1161-6.
4. Popat S, Ardizzoni A, Ciuleanu T, Cobo Dols M, Laktionov K, Szilasi M. Nivolumab in previously treated patients with metastatic squamous NSCLC: results of a European, single arm, phase 2 trial (CheckMate 171) including patients aged >=70 years or with poor Performance Status. Ann Oncol 2017;28 (Suppl 5): v460-v96.
5. Middleton G, Brock K, Summers Y, Connibear J, Shah R, Ottensmeier C. Pembrolizumab in performance status 2 patients with non-small cell lung cancer (NSCLC): results of the PePS2 trial. Ann Oncol 2018;(suppl 8) viii493-viii547.
6. Barlesi F, Audigier-Valette C, Felip E, Ciuleanu TE, Jao K, Rijavec E. CheckMate 817: first-line Nivolumab + ipilimumab in patients with ECOG PS 2 and other special populations with advanced NSCLC. 2019 World Conference on Lung Cancer in Barcelona (Spain.
7. Spigel DR, McCleod M, Jotte RM, Einhorn L, Horn L, Waterhouse DM, et al. Safety, Efficacy, and Patient-Reported Health-Related Quality of Life and Symptom Burden with Nivolumab in Patients with Advanced Non-Small Cell Lung Cancer, Including Patients Aged 70 Years or Older or with Poor Performance Status (CheckMate 153). J Thorac Oncol 2019;14(9):1628-39.
8. Passaro A, Spitaleri G, Gyawali B, de Marinis F. Immunotherapy in Non-Small-Cell Lung Cancer Patients With Performance Status 2: Clinical Decision Making With Scant Evidence. J Clin Oncol 2019;37(22):1863-7.
9. Pluvy J, Brosseau S, Naltet C, Opsomer M-A, Cazes A, Danel C, et al. Lazarus syndrome in nonsmall cell lung cancer patients with poor performance status and major leukocytosis following nivolumab treatment. Eur Respir J 2017;50(1).
10. McLoughlin EM, Larner JM, Bergman MP, Stelow EB, Brady K, Lynch AC, et al. Rapid Response to Pembrolizumab in a Critically Ill Mechanically Ventilated Patient with New Diagnosis of NSCLC. J Thorac Oncol 2019;14(9):e193-5.
11. Bonomi M, Ahmed T, Addo S, Kooshki M, Palmieri D, Levine BJ, et al. Circulating immune biomarkers as predictors of the response to pembrolizumab and weekly low dose carboplatin and paclitaxel in NSCLC and poor PS: An interim analysis. Oncol Lett 2019;17(1):1349-56.